블로그
[BLD] Fluorogenic Covalent Probes for RNA via 2′-Hydroxyl Acylation with High Selectivity
페이지 정보
조회 87회 작성일 26-01-09 16:06
본문
11 December 2025
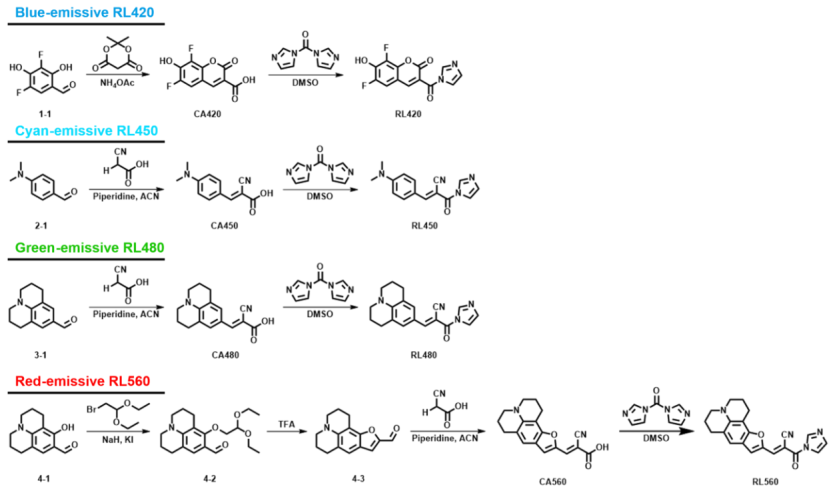
A research team led by Eric T. Kool (Stanford University) reported a novel RNA-selective fluorogenic labeling strategy in the Journal of the American Chemical Society. The study introduces RiboLight (RL) fluorophores, a new class of covalent probes that selectively label RNA by targeting the 2′-hydroxyl (2′-OH) group of the ribose backbone.
The RL probes utilize acylimidazole-mediated acylation to form a covalent ester bond with RNA, while DNA—lacking the 2′-OH—remains largely unreactive. This results in exceptional RNA selectivity, reaching up to 970-fold over DNA, along with strong fluorescence activation. The probes are designed as fluorogenic molecular rotors, remaining weakly fluorescent in solution but becoming highly emissive upon covalent attachment to RNA due to restricted intramolecular rotation.
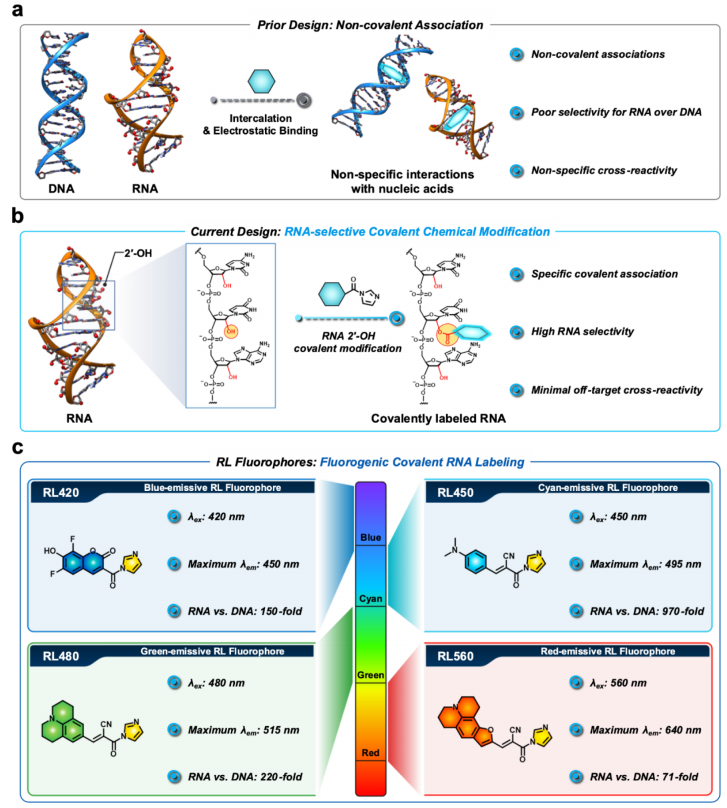
This approach enables sequence-independent RNA labeling under mild aqueous conditions, with up to 390-fold fluorescence enhancement and tunable multicolor emission. The RL platform demonstrates broad applicability, including RNA gel staining, solution-based detection, real-time RNA monitoring, and wash-free cellular RNA imaging, while exhibiting low cytotoxicity.
Overall, this work establishes the first general, covalent, and highly selective RNA-labeling platform based on 2′-OH reactivity, overcoming the limitations of conventional noncovalent RNA dyes.
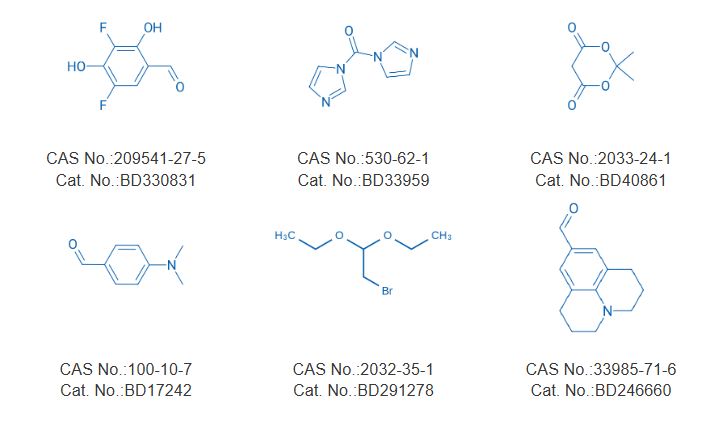
Building Blocks Suggestion
BLD Pharm supplies a comprehensive portfolio of high-purity building blocks, acylimidazoles, and dye reagents suitable for the synthesis of RL fluorophores. These materials fully support the reported synthetic routes and enable efficient development of RNA-selective fluorogenic probes for research and industrial applications.
▼ Related Building Blocks
- 이전글[Enamine] Metal Chelators for Chemotherapy 26.01.09


 카테고리 전체보기
카테고리 전체보기